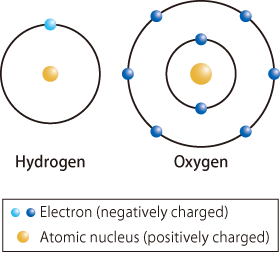
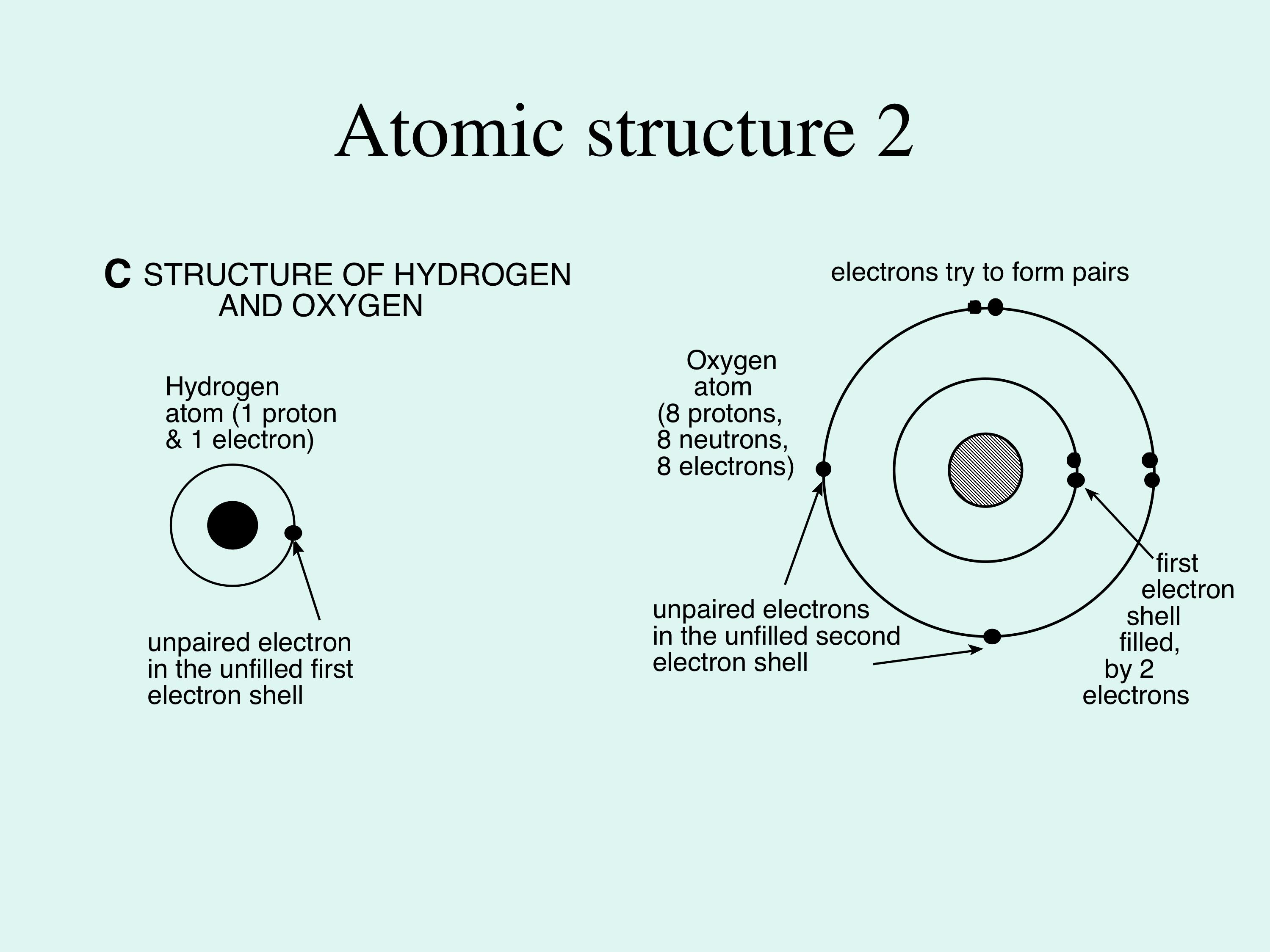
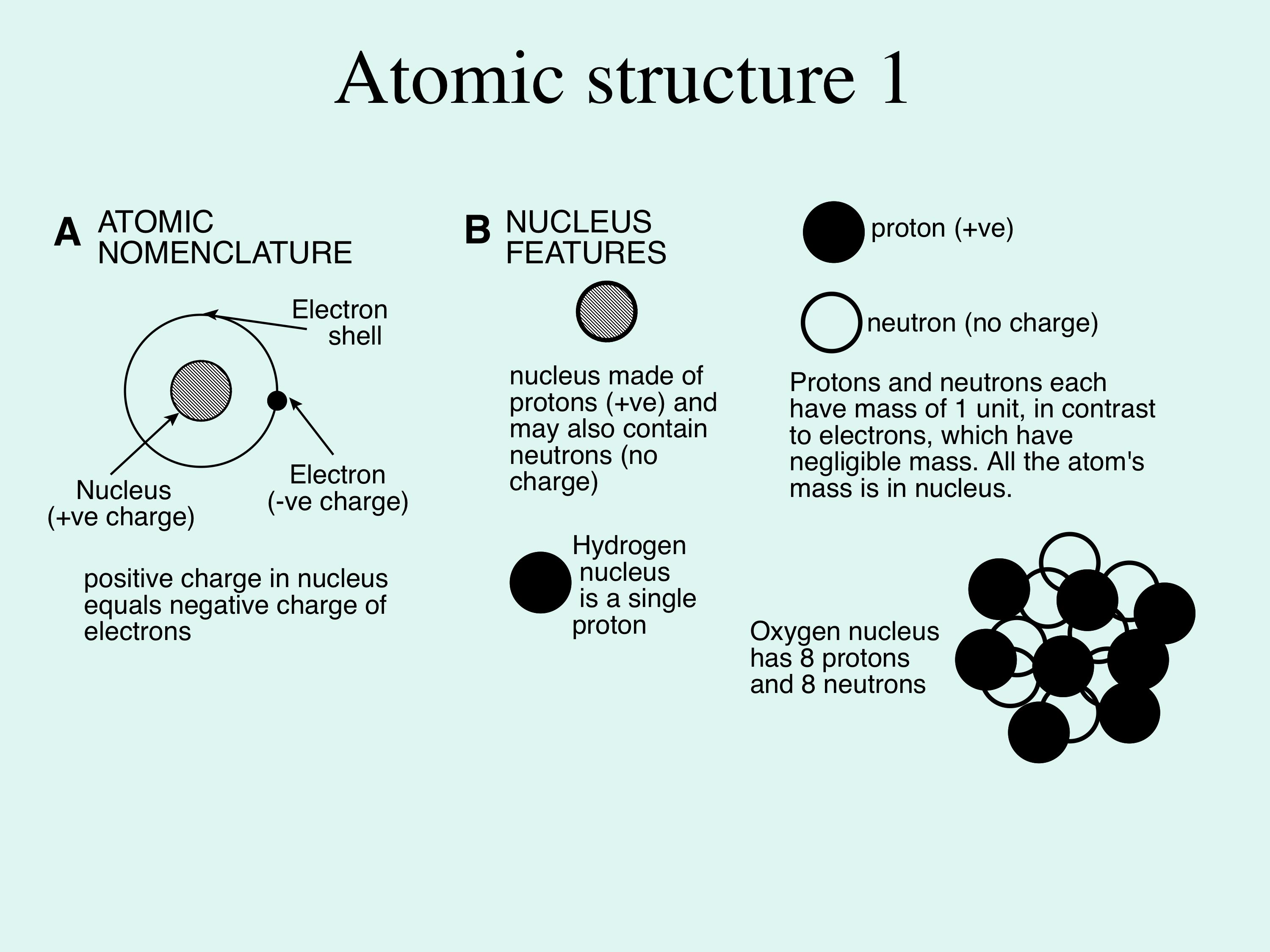
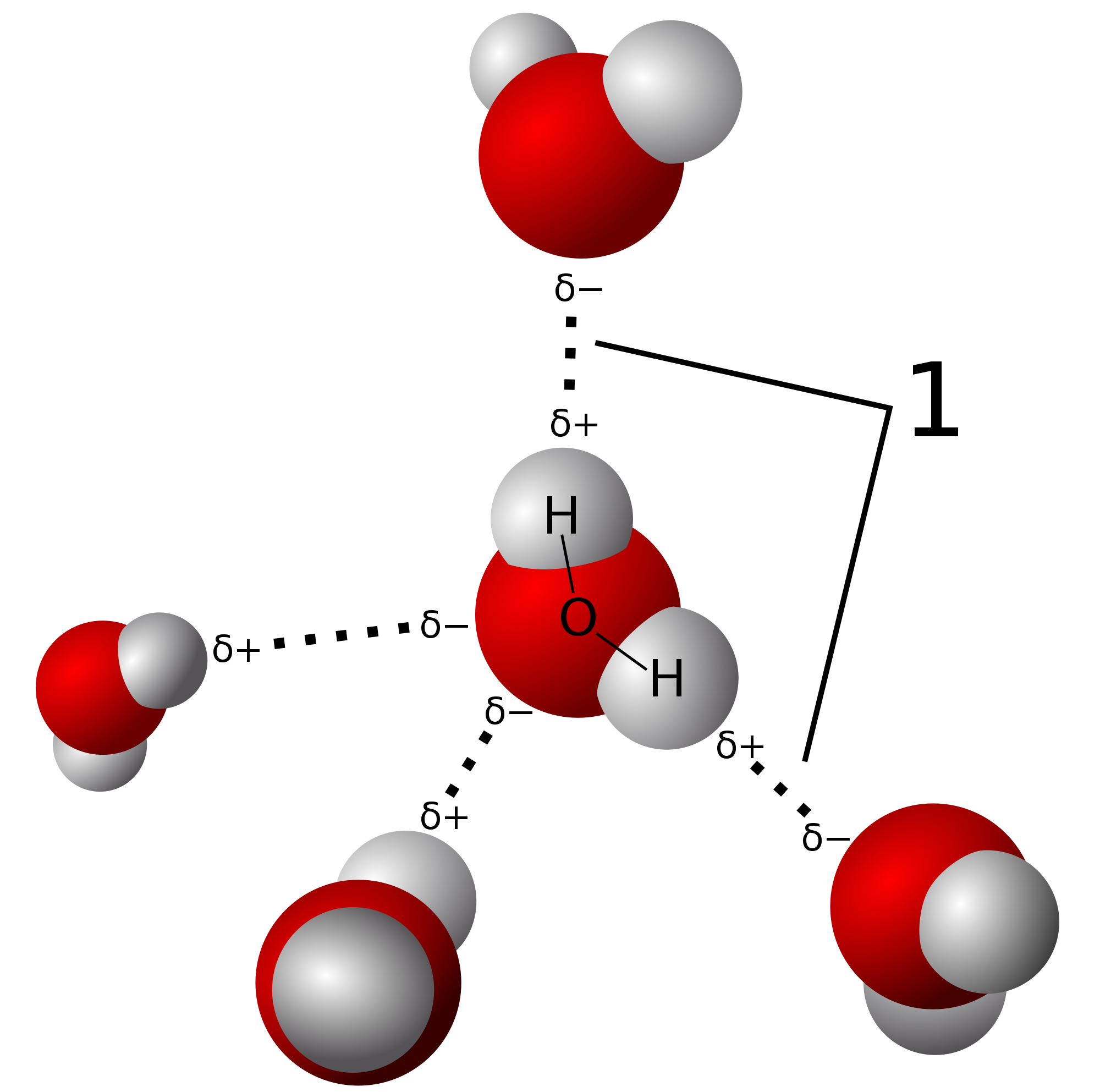
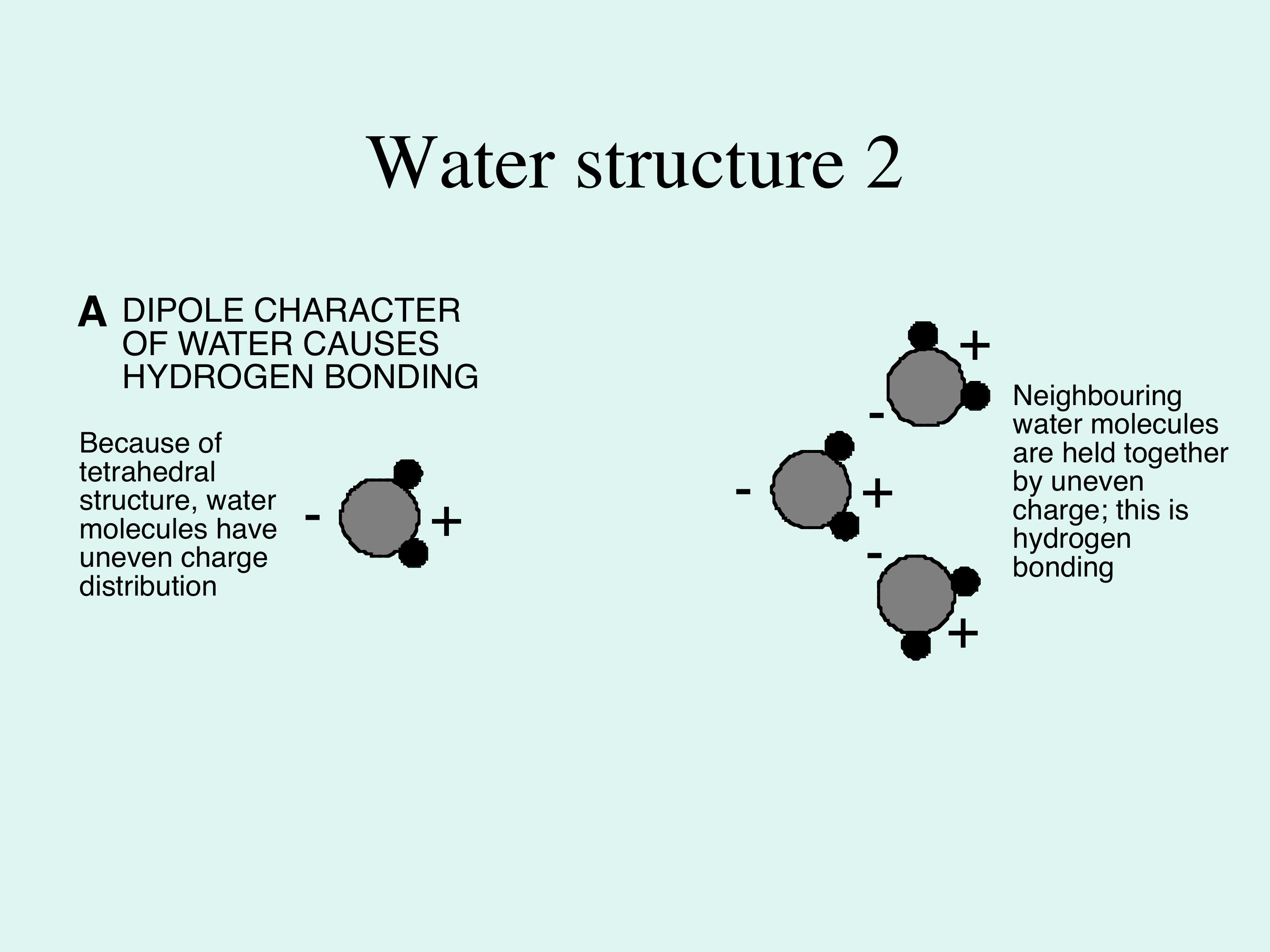
The Extraordinary Properties of Water. Water A water molecule (H 2 O), is made up of three atoms --- one oxygen and two hydrogen. The bonds between the. - ppt download

Chemistry 11Chapter 4 Chemical Bonding & Molecular Structure Hydrogen Bond -Inter & Intra Molecular - YouTube

Whose dog is this? Hydrogen peroxide, disproportionation reactions, oxidation numbers, electronegativity & more! – The Bumbling Biochemist
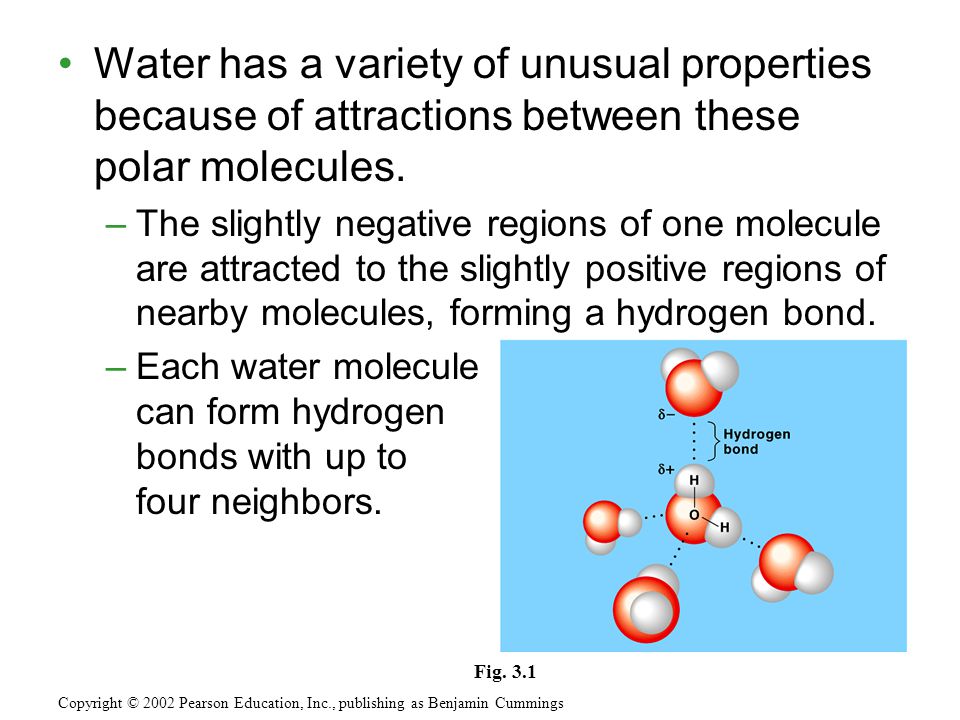
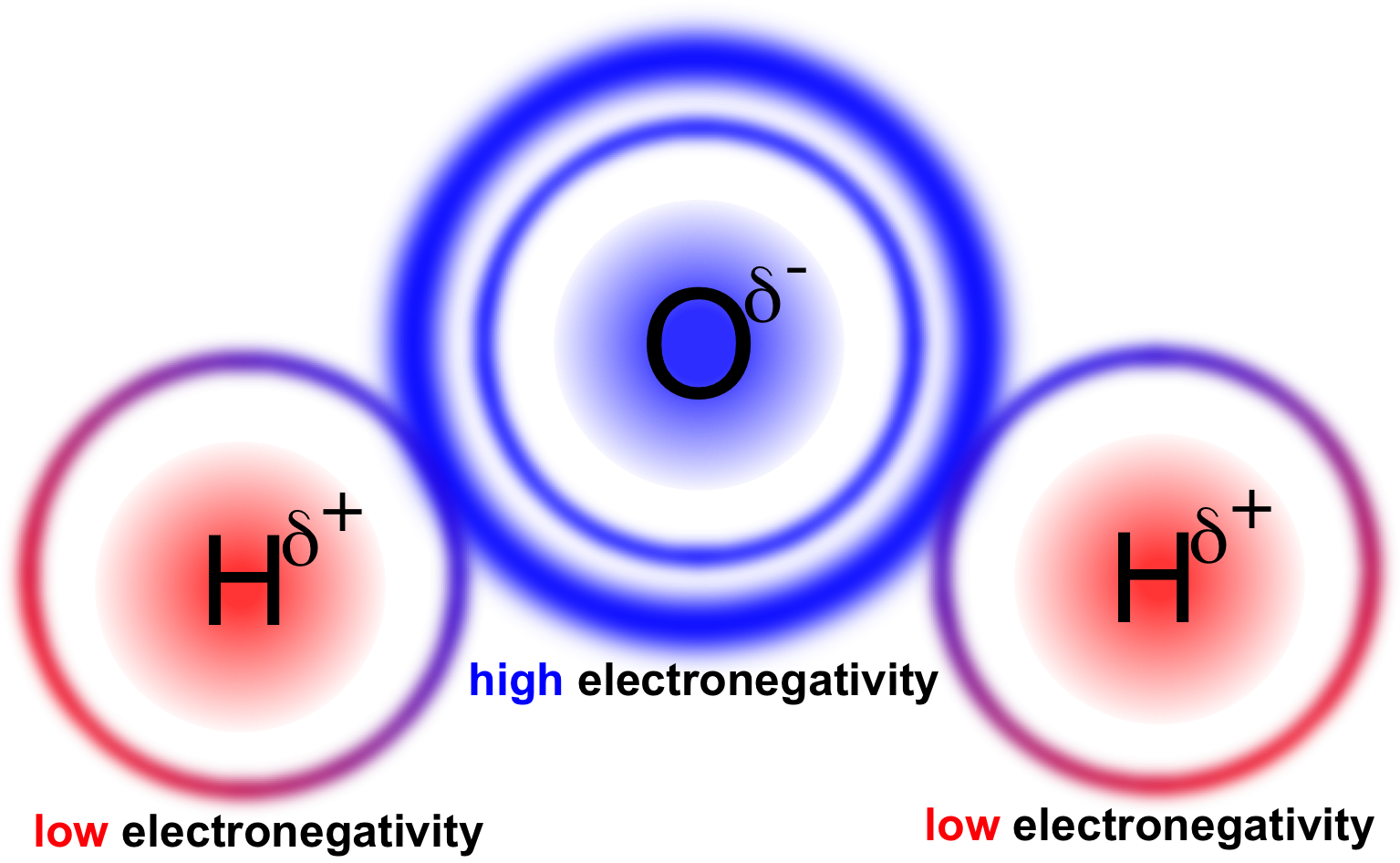
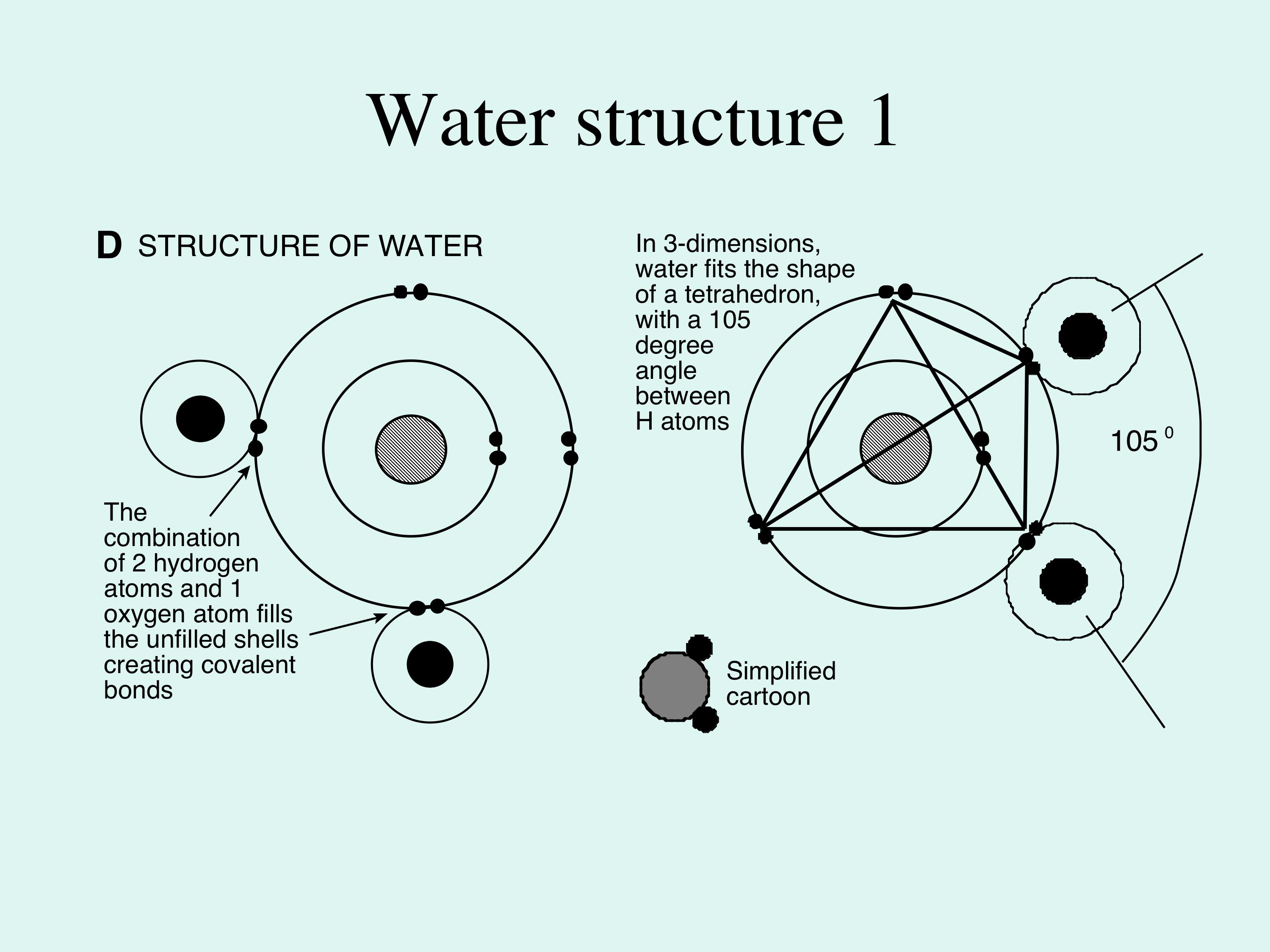
In a water molecule two hydrogen atoms form single polar covalent bonds with an oxygen atom. –Because oxygen is more electronegative, the region around. - ppt download